Deciphering the molecular mechanism of water boiling at heterogeneous interfaces

Water, with its unique thermodynamic properties, which drastically change at the state points of phase transition is omnipresent in everyday life and natural processes. Water is also one of the most widely used coolants and solvents in industrial applications. Boiling is a first-order phase transition characterized by a discontinuity of the heat capacity. In high-temperature thermo-hydraulics, the cooling of material surfaces is particularly efficient in the boiling regime as long as the surface is covered with a film of liquid like water. At these conditions, the surface heat can be efficiently removed via the liquid phase and consumed through the evaporation process. At the critical heat flux (CHF), which is the limit of the safe operating conditions of a system, the water film vaporizes (unrestrained bubble expansion parallel to the heated surface) and the surface comes into contact with the vapor phase, causing the dry-out phenomenon. The heat transfer rate is reduced and heat cannot be efficiently dissipated at such conditions, provoking a local temperature rise of the underlying surface material. The dry-out phenomenon has a devastating impact on the aging of the solid surface affecting the efficient heat removal (from solid to fluid), which potentially reduces the service life and the safe operation of industrial infrastructure. Understanding the conditions under which cavities (seeds of the vapor bubbles) prefer to form and the presence of a thin adsorbed layer is crucial for the safe operation of power plants. Based on the fundamental understanding of the nucleation mechanism, new materials (both in terms of structure and functionality) can be developed with optimized functionality (i.e., efficient surface heat removal/exchange with the coolant).
The heterogeneous water boiling mechanism on the monoclinic zirconia (used as thermal barrier coatings material, cladding material in the fuel assemblies of the nuclear power plants etc.) was investigated using the Transition Path Sampling (TPS) molecular dynamics (MD) simulations. The main advantage of the TPS approach precut in this work is that the free energy surface of vapor nucleation is obtained based on self-consistent continuous atomic trajectory tracking the full-length dynamical pathways from liquid to vapor and vice versa. On this basis, an unprejudiced (without introducing bias and overheat) investigation of the dynamics of bubble nucleation and growth is enabled. The simulations provide insight into the effect of hydrophilic and hydrophobic interactions on the boiling mechanism, the formation of a thin molecular film of adsorbed water at fluid vapor interface and the nature of the vapor nucleation surface sites controlling.
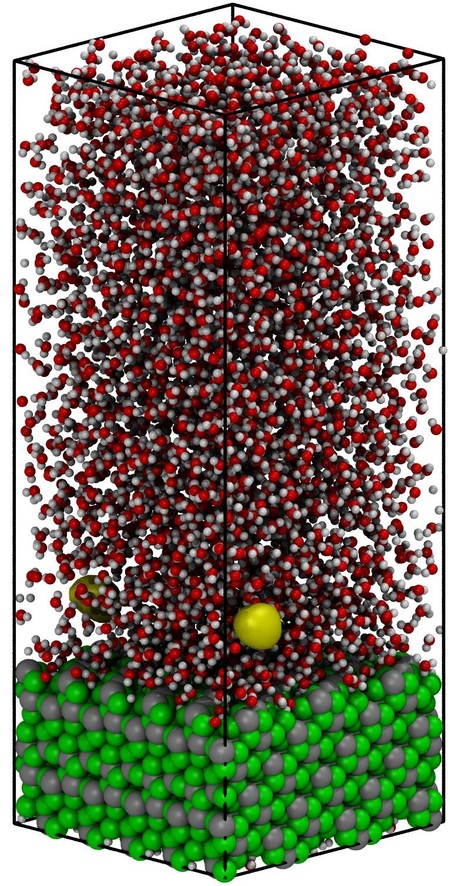
Bubble nucleation is a rare event associated with a large activation energy barrier. Spontaneous formation of the vapor cavities at the boiling temperature cannot be observed in conventional molecular dynamics (MD) simulations due to the rarity of such fluctuations and the wait time between consecutive events. Consequently, the TPS approach was applied to understand the bubble nucleation mechanism in the liquid–vapor transition at the vapor–liquid-solid interface. The TPS approach’s initial step is to generate a first, non-equilibrium trajectory leading from the liquid into the gas phase. The latter is achieved by overheating the equilibrated system from 396 to 800 K with a constant rate of 1 K/ps. During the heating, the water density decreases progressively and at a certain point of a few picosecond duration the fluid density drops from 0.6 to 0.1 g/cm3. This sharp density drop is characterized as explosive (massive) boiling in which complex-shaped randomly distributed cavities (bubbles) emerge in the simulation domain.
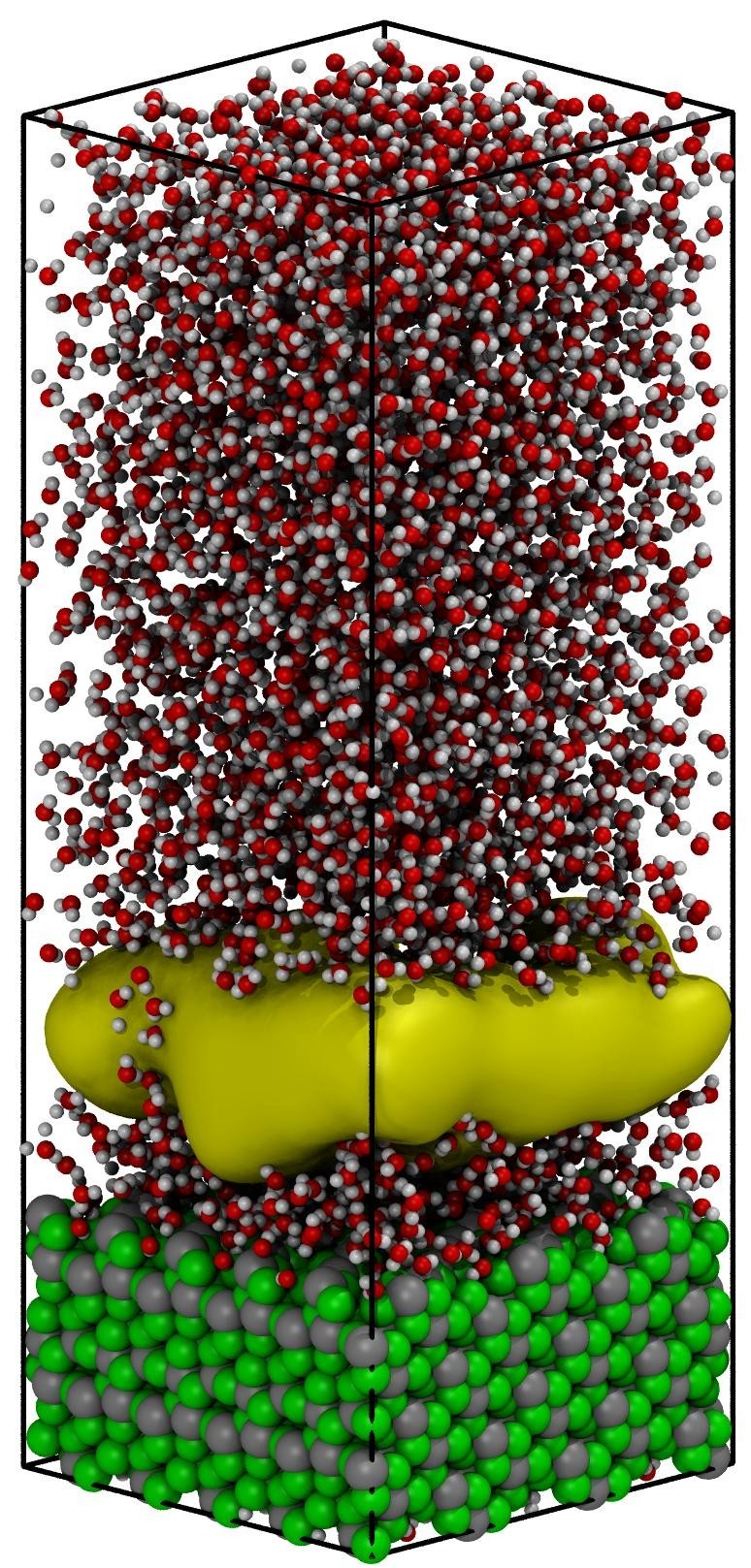
Figure 1 shows a representative set of snapshots illustrating the vapor nucleation’s consequent steps and boiling at a constant temperature. Based on these configurations, the heterogeneous water boiling mechanism at liquid–vapor transition temperature (100oC) and the nucleation location was analyzed. Cavities (diameter higher than 3 Å) are absent at liquid-like densities of 0.9 g/cm3 (Fig. 1a). After several hundreds of picoseconds, multiple small cavities are formed in bulk. The formed cavities break the hydrogen-bonded network of the bulk water and act as a trigger for the formation of more cavities, which progressively merge and create a large vacuum domain (Fig. 1d). Analysis of the nucleation center distribution shows that the highly hydrophilic solid interface is covered with a thin film of water molecules. The cavities do not nucleate at the interface between the solid and liquid phase but between the ordered film of water at the surface and bulk like water located ~ 7 Å away from the solid surface.
In the first stages of the boiling process, subcritical vapor bubbles (cavities with a radius smaller than 5 Å) are formed in the liquid domain and subsequently disappeared (condensate). At steady-state conditions, all bubbles with a radius smaller than 10 Å are expected to collapse since the Young–Laplace pressure is smaller than the total pressure of the system. Consequently, the TPS approach allowed us to visualize tiny radius cavities’ nucleation for the first time suggesting that the transition from the liquid to the vapor phase is triggered by nucleation and subsequent growth of the vapor phase seeds.
 |
 |
 |
 |
Original Publications
Konstantinos Karalis, Dirk Zahn, Nikolaos I. Prasianakis, Bojan Niceno & Sergey V. Churakov (2021). Deciphering the molecular mechanism of water boiling at heterogeneous interfaces, Scientific Reports.
Prof. Dr. Sergey Churakov
PSI
sergey.churakov@psi.ch
Dr. Konstantinos Karalis
PSI
konstantinos.karalis@psi.ch